In thermodynamic systems, there is conversion of law of energy that is associated with most of the systems. While dealing with law of this system, it is noteworthy that certain issues be taken into consideration.
The law of conservation of energy is used for knowing what total energy an isolated system has and how energy can be transferred and transformed from one segment to another without wasting it in any manner.
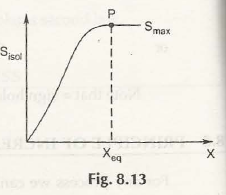
In this case, entropy has an important role to play, wherein it is maximum at the equilibrium level while it increases when placed in a secluded system.
Consider the following figure:
Links of Previous Main Topic:-
- Open system and control volume
- Conversion of work into heat
- Introduction to carnot cycle
- Clausius inequality entropy and irreversibility introduction
- Reversible adiabatic paths do not intersect
- Clausius theorem
- Entropy
- Entropy temperature plot
- Clausiuss inequality
- Entropy change in an irreversible process
- Principle of increase of entropy
- The degree of irreversibility of and irreversible process
- Summary of first and second law by clausius
Links of Next Mechanical Engineering Topics:-
- Practical use of entropy principle
- First law and second law combined
- Analysis of thermodynamic equations
- Ideal gas or perfect gas
- Introduction about air standard cycles
- Properties of pure substances introduction
- Vapour compression refrigeration cycle introduction
- Basic fluid mechanics and properties of fluids introduction