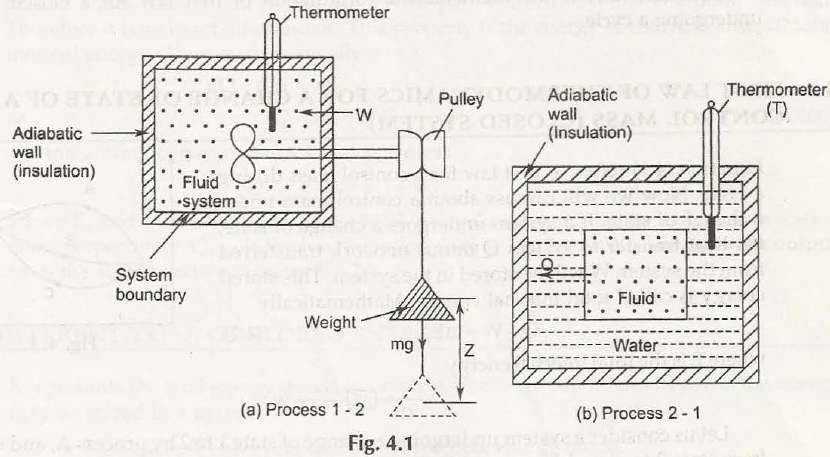
A system performs through a cycle that is made of two different processes. In case of first process it would work on system through paddling which will help turn only if the weight is lowered. If the system returns to initial state then transfer heat from one system until cycle gets completed.
The work is measured through mechanical units of force times distance and it is measured through joules and foot pounds force and heat would finally be measured through thermal units that are symbolized as British thermal unit (Btu).
The work and heat measurement is done only during a cycle for different types of system and for various amounts of heat and work. While the amount of heat and work compared, it is visualized that they appears to be proportional. This observation helps us to retrieve the formula related to first law of thermodynamics:
| (Eq1) |
|
First law of thermodynamics for closed system undergoing a cyclic process
The transfer of heat as well as performance of work in both cases there will be the same effect in system. Energy would get into system as heat way leave the system as work and energy would has entered as work would transfer into heat. The law of conservation of energy would say that the net work performed by system is equal to that of net heat supplied to system.
When any system goes through a thermodynamics cyclic process, then the heat get supplied to system from surrounds which is equal to work done by system on surroundings.
Links of Previous Main Topic:-
- Introduction to statics
- Introduction to vector algebra
- Two dimensional force systems
- Introduction concept of equilibrium of rigid body
- Friction introduction
- Introduction about distributed forces
- Area moments of inertia in rectangular and polar coordinates
- Mass moment of inertia introduction
- Work done by force
- Kinematics of particles
- Position vector velocity and acceleration
- Plane kinematics of rigid bodies introduction
- Combined motion of translation and rotation
- Rectilinear motion in kinetics of particles
- Work and energy
- Linear momentum
- Force mass acceleration
- Simple stress introduction
- Normal strain
- Statically indeterminate system
- Introduction to thermodynamics
- Statement of zeroth law of thermodynamics with explanation
- Heat and work introduction
Links of Next Mechanical Engineering Topics:-
- First law of thermodynamics for a change of state for a control mass closed system
- Different types of stored energy
- The constant volume specific heat
- Enthalpy
- The constant pressure specific heat
- Specific heats of solids and liquids
- Energy of isolated system
- Perpetual motion machine of the first kind pmm 1