The measurable characteristics or the substances that can be premeditated is calledthe property of the system. Pressure, volume and temperature are the best examples of macroscopic nature properties. The features areusually independent of mass, pressure, temperature and volume are known as intensive properties.
Extensive properties are those properties which are not independent of volume, mass and energy.
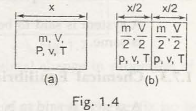
In the above figure, 1.4(a) represents a substance with mass(m), volume(v) and temperature(T) to which now a diaphragm is set to the middle of the container. Now we obtain two chambers that are represented in fig 1.4(b. The two chambersconsist of volume V/2, mass m/2 keeping pressure and temperature as constant. Therefore, V accounts for thelarge property and P, T and v account for the intensive properties of the material.
1.6.2 State
The condition of the system when it has certain properties and then expressed at the time when the thermodynamic property coordinates at the points (P,v) coordinate.
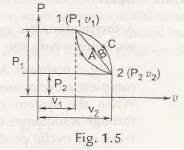
In the above fig 1.5, it accounts for apointone which is at the condition (P1 v1) and points 2 at (P2 v2). When any changes occur from state 1 to state 2, there will be some heat or work transfer will be happening. The dissimilarity between the coordinates of the two points that shows the states is the change that occurs with the absolute value of the property in the state 1 and state 2.
ΔP = P2 – P1 , and Δv = v2 –v1
In the above equation (1.1) it represents that the possessions do not be determined by the system when the changes occur and it depends on the state. Thus the variations in the properties of the paths A,B,C do not affect at the end point(1 and 2). Thus these properties are called the point functions. It is considered as property when any value is in a state of equilibrium.
Links of Previous Main Topic:-
- Linear momentum
- Force mass acceleration
- Simple stress introduction
- Normal strain
- Statically indeterminate system
- Introduction to thermodynamics
- Macroscopic and microscopic point of view
Links of Next Mechanical Engineering Topics:-
- Thermodynamic equilibrium
- Processes cycles
- Quasi static process
- 1 10 1 reversible process
- Statement of zeroth law of thermodynamics with explanation
- Heat and work introduction
- First law of thermodynamics for a control mass closed system undergoing a cycle
- Open system and control volume
- Conversion of work into heat