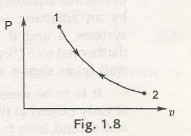
When the process is entirely reversed, then it is called a reversible process. In the graph shown below figure 1.8, we could see a process moving in the forward direction from 1 to 2, and this process is carried out in the reverse direction that is fro 2 to 1. This way it again comes back to its first state. This system of returning into its original version is called a reversible process. Thus the interaction between the system and surrounding might be same but is in the opposite direction. This makes the surroundings also to come to its originalstate. Thus it can be concluded the quasi-static system is a reversible process.
1.10.2 Irreversible Process
That system which cannot be changed is called an irreversible process. An example can be sorted out with a quasi-static process with the system as an irreversible process. The process which is in a state of nonequilibriumis those who cannot be reversed. Because in this system it cannot be brought into the original state without leaving any change in the environment.
Links of Previous Main Topic:-
- Linear momentum
- Force mass acceleration
- Simple stress introduction
- Normal strain
- Statically indeterminate system
- Introduction to thermodynamics
- Macroscopic and microscopic point of view
- Concepts of systems
- Control volume and control surface
- Homogeneous and heterogeneous systems
- 1 6 1 property
- Thermodynamic equilibrium
- Processes cycles
- Quasi static process
Links of Next Mechanical Engineering Topics:-