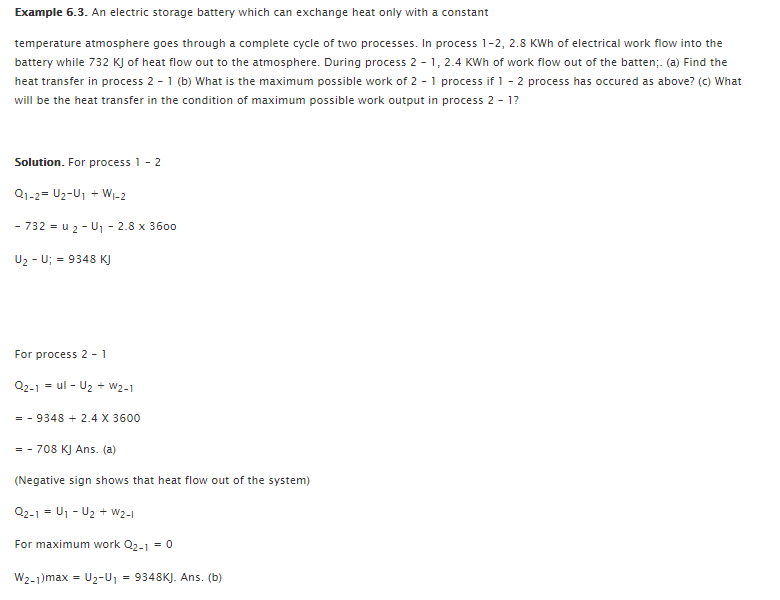
There are two different statements related to second law of thermodynamics:
- Kelvin Plank statement
- Clausius statement
Clausius statement of thermodynamics
This is impossible for any self acting machine which may work in cyclic process and unaided by external agency that helps to convey heat energy from any object that comes with lower temperature to object at higher temperature.
Kelvin Plank statement
This appears to be impossible which can construct any engine which might help while operating in any cycle that can produce no effect which can except to retrieve any heat energy from any single thermal energy reservoir and it is equivalent to amount of work.
Equivalence of Kelvin Plank and Clausius statement
As per Kelvin Plank and Clausius statement, it is found that both statements differ with each other. But, it is important to analyze these two statements and come to a conclusion that both these statements are equivalent to each other.
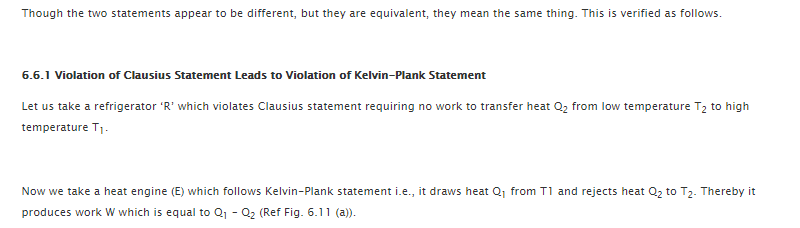
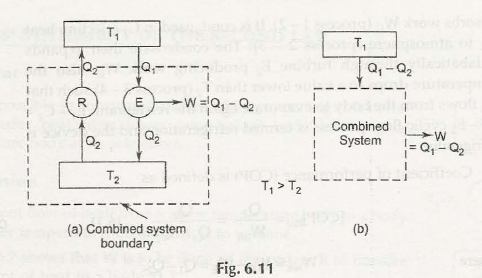
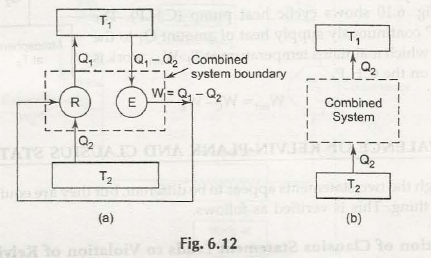
So, let us prove that second law of thermodynamics is equivalent statement in all respects. This is possible by reflecting that violation of any one statement would certainly violate the other statement and vice versa.
In case the thermodynamic system is not able to follow Kelvin Plank statement then it proves that it would neither follow the Clausius statement. The same thing happens when Clausius statement is not followed.
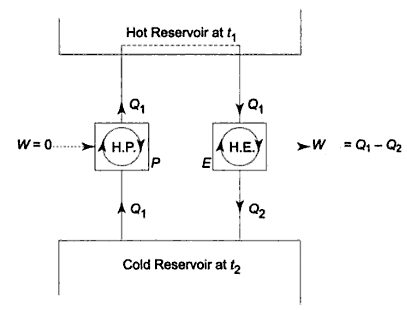
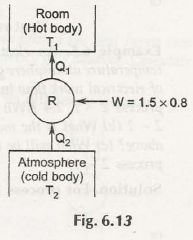
The figure shows that heat pump is transferring heat energy Q1 from any lower temperature body to higher temperature body that does not have any other effect. This means that heat pump would receive heat energy Q1 that is obtained from lower temperature body and finally gets delivered into Q1 to any higher temperature body and this does not secure any kind of work energy from surrounding.
Therefore, we can say that Clausius statement is not followed by system and clausius statement can get violated.
Links of Previous Main Topic:-
- Statically indeterminate system
- Introduction to thermodynamics
- Statement of zeroth law of thermodynamics with explanation
- Heat and work introduction
- Open system and control volume
- Conversion of work into heat
- Kelvin plank statement of second law of thermodynamics
- Clausius statement of the second law
Links of Next Mechanical Engineering Topics:-